Quality Assurance (QA)
What is Quality Assurance (QA)?
Quality Assurance (QA) is a way of preventing mistakes and defects in manufactured products and avoiding problems when delivering products or services to customers; which ISO 9000 defines as "part of quality management focused on providing confidence that quality requirements will be fulfilled". This defect prevention in quality assurance differs subtly from defect detection and rejection in quality control and has been referred to as a shift left since it focuses on quality earlier in the process (i.e., to the left of a linear process diagram reading left to right). Quality assurance comprises administrative and procedural activities implemented in a quality system so that requirements and goals for a product, service, or activity will be fulfilled. It is the systematic measurement, comparison with a standard, monitoring of processes, and an associated feedback loop that confers error prevention. This can be contrasted with quality control, which is focused on process output.
Quality assurance includes two principles:
- "Fit for purpose" (the product should be suitable for the intended purpose); and
- "right first time" (mistakes should be eliminated).
QA includes management of the quality of raw materials, assemblies, products and components, services related to production, and management, production, and inspection processes. The two principles also manifest before the background of developing (engineering) a novel technical product: The task of engineering is to make it work once, while the task of quality assurance is to make it work all the time. Historically, defining what suitable product or service quality means has been a more difficult process, determined in many ways, from the subjective user-based approach that contains "the different weights that individuals normally attach to quality characteristics," to the value-based approach which finds consumers linking quality to price and making overall conclusions of quality based on such a relationship[1]
How Quality Assurance Works[2]
Quality assurance methods focus on establishing good processes to produce products with the quality already built-in, rather than going through an unmonitored production process and trying to “inspect the quality” of a product that’s already been finished. Concepts of quality control can be traced back to at least the Middle Ages and the rise of guilds. A craftsman could access a network of connections with other craftsmen and suppliers by joining a guild organization. He could then benefit from the reputation of the guild based on standards of quality in the products produced by its members. The Industrial Revolution brought about more specialization in labor, as well as mechanization. Quality assurance evolved to address specialized tasks performed by workers. With the introduction of mass production, the need to monitor the quality of components being produced by large numbers of workers created a role for quality inspectors.
Modern Approaches
Today's approaches to quality assurance may differ depending on the industry. For example, a hospital might implement QA methods to improve the quality of healthcare. This could include defining quality by understanding core activities in effective patient care, setting quality benchmarks, and measuring quality with surveys, audits, and supervision. Another business, such as a nutritional supplement manufacturer, would have completely different methods to ensure quality since its product parameters are completely different than a hospital. For example, the supplement company might guide its processes based on guidelines for active pharmaceutical ingredients.
International Quality Assurance Standards
The International Organization for Standardization (ISO) was founded in 1947 with the goal of ensuring quality across national borders.1 ISO consists of standards organizations that represent over 160 countries. It maintains an effective quality assurance system for manufacturing and service industries. One product of the ISO is the set of standards that have become known as the ISO 9000 family. The criteria detailed in these management systems are designed to help organizations meet statutory and regulatory requirements for product quality and consumer needs. Manufacturers and businesses can become certified by fulfilling the requirements defined in ISO 9001. This offers independent confirmation of an organization's adherence to quality standards. More than 1 million organizations around the world are ISO 9001 certified. To obtain ISO 9001 certification, the management of an organization develops quality assurance goals that are codified into policies and guidelines. These policies and guidelines are specifically designed to meet the unique needs of its business and manufacturing processes, and they're often crafted with the assistance of a consultant. Once the organization implements the guidelines, the systems are assessed for compliance with ISO 9000 standards. The results of the assessment identify any areas that fall outside of ISO standards. If the organization addresses any problem areas within a given time frame, then it receives certification.
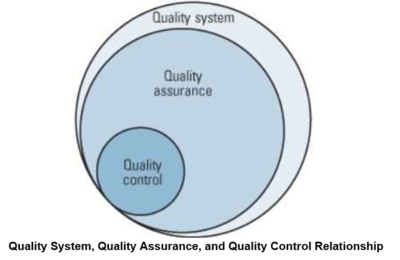
Quality assurance Vs. Quality Control[3]
Quality assurance and quality control are two aspects of quality management. While some quality assurance and quality control activities are interrelated, the two are defined differently. Typically, QA activities and responsibilities cover virtually all of the quality system in one fashion or another, while QC is a subset of the QA activities. Also, elements in the quality system might not be specifically covered by QA/QC activities and responsibilities but may involve QA and QC. The Figure below shows ISO 9000 definitions from ISO 9000:2015: Quality management systems - Fundamentals and Vocabulary.
Quality Assurance
Quality assurance can be defined as "part of quality management focused on providing confidence that quality requirements will be fulfilled." The confidence provided by quality assurance is twofold—internally to management and externally to customers, government agencies, regulators, certifiers, and third parties. An alternate definition is "all the planned and systematic activities implemented within the quality system that can be demonstrated to provide confidence that a product or service will fulfill requirements for quality."
Quality Control
Quality control can be defined as "part of quality management focused on fulfilling quality requirements." While quality assurance relates to how a process is performed or how a product is made, quality control is more the inspection aspect of quality management. An alternate definition is "the operational techniques and activities used to fulfill requirements for quality."
Industry Perspectives on QA and QC
For some service organizations, the concept of quality control may be foreign because there is no tangible product to inspect and control. The quality assurance function in a service organization may not include quality control of the service but may include quality control of any products involved in providing the service. A service may include products that are documents (such as a report, contract, or design) or tangible products (such as a rental car or units of blood). It may be necessary to control product quality in a service organization to ensure that the service meets customer requirements.
History of QA and QC
Quality has been defined as fitness for use, conformance to requirements, and the pursuit of excellence. Even though the concept of quality has existed from early times, the study and definition of quality have been given prominence only in the last century.
- 1920s: Quality Control: Following the Industrial Revolution and the rise of mass production, it became important to better define and control the quality of products. Originally, the goal of quality was to ensure that engineering requirements were met in the final products. Later, as manufacturing processes became more complex, quality developed into a discipline for controlling process variation as a means of producing quality products.
- 1950s: Quality Assurance and Auditing: The quality profession expanded to include quality assurance and quality audit functions. The drivers of independent verification of quality were primarily industries in which public health and safety were paramount.
See Also